
가이드라인 개발에 평균 2년 이상이 소요되었다는 점을 배울만하다. 서두른다고 좋은 결과가 나오는 것은 아니다.
[20110821. guideline (10): SIGN50 - a guideline developer's handbook]
SIGN50은 스코트랜드의 가이드라인 개발자를 위한 가이드라인 같은 것이다. 2008년 1월에 revised version이 발표되었다. 총 10개의 chapter로 되어 있다. 맨 첫 페이지는 SIGN methodology가 AGREE appraisal criteria에 따른다는 점을 밝히고 있다. 그만큼 AGREE가 중요하다.

가이드라인 개발에 평균 2년 이상이 소요되었다는 점을 배울만하다. 서두른다고 좋은 결과가 나오는 것은 아니다.
[Chapter 1. Introduction]
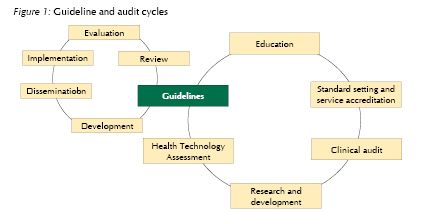
1.4. Medico-legal implications: The Hunter v Hanley test applied by the Court is based on what is actually done in practice rather than on a prescription of what should be done as proposed by guidelines. It is important to emphasise that SIGN guidelines are intended as an aid to clinicl judgement not to replace it. Practitioners should all be able to define the standard of care which they provide, and to justify if necessary why these do not meet nationally agreed recommendations.
[Chapter 2. Organisation of guideline development]
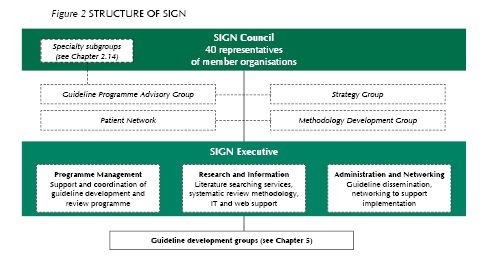
[Chapter 3. Selection of guideline topics]

[Chapter 4. Involving patients and their representatives]
[Chapter 5. The guideline development group]
The life span of each guideline development group is approximately 28 months, with groups meeting on average once every two months, although groups may form subgroups which meet more frequently. Guideline development groups are supported by the SIGN Executive.
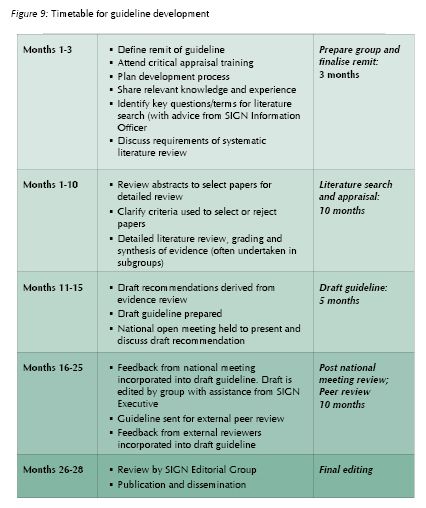
[Chapter 6. Systematic literature review]
Members of SIGN guideline development groups are encouraged to break down the guideline remit into a series of structured key questions using the PICO format (Patients or populations, Intervention, Comparision, Outcome).
Deciding the key questions is entirely the responsibility of the guideline development group
Key questions form the basis of the literature search, which is undertaken by a SIGN Information Officer.
[Chapter 7. Forming guideline recommendations]
Evidence tables are compiled by SIGN executive staff based on the quality assessments of individual studies provided by guideline development group members.

Finally, the group is asked to consider the importance of the recommendation(s) it has just made. Importance is not necessarily related to strength of evidence, but should reflect the extent to which the group believes the recommendation will impact on the health status or quality of life of the patients concerned.
[Annex to chapter 7. New grading system introduced in 2000]
[Levels of evidence]
1++: High quality meta-analyses, systematic reviews of RCTs, or RCTs with a very low risk of bias
1+: Well-conducted meta-analyses, systematic reviews, or RCTs with a low risk of bias
1-: Meta-analyses, systematic reviews, or RCTs with a high risk of bias
2++: High quality systematic reviews of case control or cohort or studies
High quality case control or cohort studies with a very low risk of confounding or bias and a high probability that the relationship is causal
2+: Well-conducted case control or cohort studies with a low risk of confounding or bias and a moderate probability that the relationship is causal
2-: Case control or cohort studies with a high risk of confounding or bias and a significant risk that the relationship is not causal
3: Non-analytic studies, e.g. case reports, case series
4: Expert opinion
[Grades of recommendations]
A: At least one meta-analysis, systematic review, or RCT rated as 1++, and directly applicable to the target population; or a body of evidence consisting principally of studies rated as 1+, directly applicable to the target population, and demonstrating overall consistency of results
B: A body of evidence including studies rated as 2++, directly applicable to the target population, and demonstrating overall consistency of results; or Extrapolated evidence from studies rated as 1++ or 1+
C: A body of evidence including studies rated as 2+, directly applicable to the target population and demonstrating overall consistency of results; or extrapolated evidence from studies rated as 2++
D: Evidence level 3 or 4; or extrapolated evidence from studies rated as 2+
[Good practice points]
On occasion, guideline development groups find that there is an important practical point that they wish to emphasise but for which there is not, nor is there likely to be, any research evidence. This will typically be where some aspect of treatment is regarded as such sound clinical practice that nobody is likely to question it (it could be regarded as “clinical common sense”). These are shown in the guideline as Good Practice Points (GPP).
[Chapter 8. Consultation and peer review]
[Chapter 9. Presentation and dissemination]
[Chapter 10. Implementation]
[Home]