 EndoTODAY 내시경 교실
EndoTODAY 내시경 교실
Beginner | ESA | Schedule | OPD
Seminars | Atlas | Recent | Links
 [2014 Yonsei Gastric Cancer Symposium]
[2014 Yonsei Gastric Cancer Symposium]
2014년 11월 22일 연세암센터 지하 3층 서암강당에서 열린 Yonsei Gastric Cancer Symposium에 다녀왔습니다. 위암 치료의 첫 단추부터 최근의 molecular thing까지 커버되는 흥미로운 시간이었습니다. 좋은 심포지엄을 준비한 연세대 위암팀 여러분께 감사말씀 전합니다.
 1. History of gastric cancer (연세대학교 외과 김충배)
1. History of gastric cancer (연세대학교 외과 김충배)
1881년에 비엔나의 Theodor Billroth가 pyloric obstruction 환자에서 subtotal gastrectomy를 하였고 환자가 4개월 생존한 것이 역사의 시작입니다. Billroth는 음악애호가였고 브람스의 친구였다고 합니다.
1942년 일본의 Kajitani가 lymph node dissection을 최초로 시행하였습니다. 일본에서는 1962년에 D2 dissection을 표준술식으로 확립했습니다. 우리나라에는 1970년에 D2 표준술식이 도입되었습니다. 2001년 3월 위암치료 가이드라인 제 1판이 나왔습니다. 2014년에 새 가이드라인이 나왔습니다.
우리나라에서는 세브란스병원의 Ludlow교수가 China Medical Journal에 발표한 Carcinoma in Korea 논문이 영문 첫 논문인 것 같습니다.
1993년 11월 6일 대한위암연구회가 창립되었습니다.
 2. History of Chemotherapy (연세대학교 정현철)
2. History of Chemotherapy (연세대학교 정현철)
1) Adjuvant chemotherapy
1957년 circulating tumor cell에 대한 3개의 논문이 나오면서 시작되었습니다. 이후 많은 연구가 있었으나 2000년대 초반까지의 연구에는 디자인에 문제가 많았습니다. 즉 adjuvant chemotherapy의 역할이 없는 것이 아니라 연구 디자인에 문제가 있어 입증되지 못했을 뿐입니다. 방영주, 노성훈 교수님 등의 CLASSIC trial 후 adjuvant chemotherapy에 대한 많은 좋은 연구가 나왔습니다.
2) Palliative chemotherapy
Old generation 약제의 response rate는 10-35% 정도였습니다. New generation 약제의 response rate는 50% 정도로 향상되었습니다. 그러나 아직 median survival이 1년이 넘은 연구는 적습니다. 서구보다 동양의 생존율이 약간 더 높습니다. 동양에서는 disease progression 후 생존기간이 더 깁니다. 아마 2차, 3차 치료가 활발히 이뤄지기 때문입니다.
3) Target agent
Anti-HER2: 첫 연구(TOGA trial, 2010 Lancet)의 survival gain이 2.7개월 이후 많은 target이 제시되었습니다. Target을 하나씩 검토하려면 시간이 많이 지나므로 최근에는 Multi-target multi-arm trial, umbrella protocol로 가고 있습니다.
과거에는 약제의 효과를 살피는 방향이었다면 최근에는 algorithm의 효과를 보는 방향으로 연구가 진행되고 있습니다.
 3. History of Radiotherapy (삼성서울병원 임도훈)
3. History of Radiotherapy (삼성서울병원 임도훈)
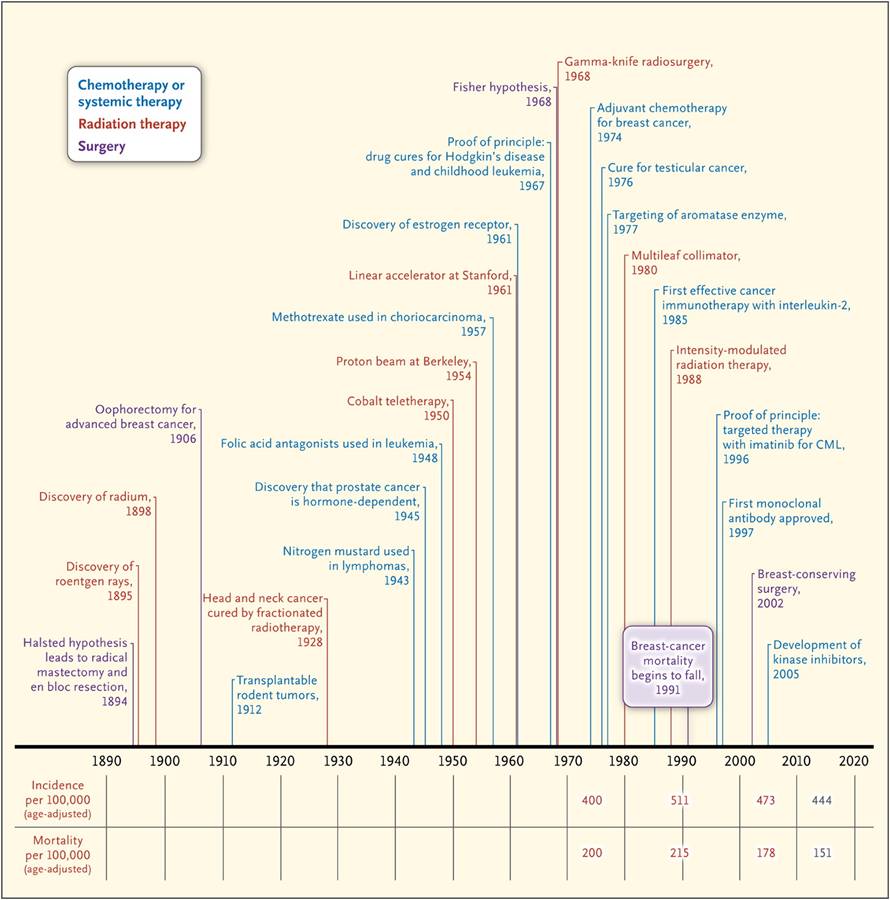
암 치료의 주요 역사 (DeVita NEJM 2012)
방사선 치료를 위해서는 pattern of recurrence에 대한 자료를 바탕으로 RT field를 잡아야 합니다. D2 dissection을 하더라도 간혹 local lymph node 혹은 dissection field 바깥쪽에서 재발하는 예가 있으므로 RT를 통하여 재발을 줄일 수 있습니다.
Pattern of recurrence at YMC (Radiother Oncol. 2012)
 4. Surgical conception on stage IV gastric cancer (Jia-fu Ji, Beijing Cancer Hospital, China)
4. Surgical conception on stage IV gastric cancer (Jia-fu Ji, Beijing Cancer Hospital, China)
2012년 전 세계 위암의 42.5%가 중국에서 진단되었습니다.
2017년 국제위암학회가 중국 베이징에서 열린다고 합니다.
 5. Molecular information based personaized drug selection ingastric cancer (연세대학교 메디컬융합연구소 박은성 박사)
5. Molecular information based personaized drug selection ingastric cancer (연세대학교 메디컬융합연구소 박은성 박사)
많은 cell line에서 조사해보면 cytotoxic drug은 효과가 있는 cell line이 많지만, targeted agent는 일부 cell line에서만 효과가 있습니다.
KRAS mutation은 위암의 5%에서 발견됩니다. 이 중 일부는 Copy number gain이 있는데 이는 MEK inhibitor 저항성의 원인이 될 수 있습니다.
강의가 너무 어려웠습니다. 내가 무식한 것인지, 강사가 강의를 못한 것인지... 잠도 오지 않아 참 곤혹스러웠습니다. 여하튼 불편했습니다.
 6. Moving forward with targets in gastric cancer from the "Sequence" to clinical trials (삼성서울병원 이지연)
6. Moving forward with targets in gastric cancer from the "Sequence" to clinical trials (삼성서울병원 이지연)
Patient-derived cell이 환자를 대표할 수 있다.
Clinical load가 많은 상황에서 개별 환자의 유전정보를 활용한 진료를 위해서는 별도의 시스템이 필요합니다. 바쁜 외래에서는 어렵습니다. 삼성서울병원에서는 개인 맞춤 암진료 클리닉을 열어 지난 8월까지 428명을 등록할 수 있었고 상당히 많은 환자에서 유전자 profile을 확인하였다고 합니다. 문제는 약이 부족하다고 합니다. 위암에서 쓸 수 있는 targeted agent가 더 많아야겠다고 판단됩니다.
이지연 선생님은 MUO환자에서 유전자 profile을 통하여 BRAF 변이가 확인되어 해당 약을 써서 dramatic하게 좋아진 환자를 보여주셨습니다.
 7. AVATAR mouse for gastric cancer (삼성서울병원 최민규)
7. AVATAR mouse for gastric cancer (삼성서울병원 최민규)
위암에서 표적 분자치료 성적의 향상을 위해서는?
1) 위암은 heterogeneous하며 암의 조직학적, 역학적, 분자학적 종류에 따라 표적분자의 발편, 증폭의 양상이 다르며 이런 점이 표적분자치료제의 반응 차이를 유발할 것이다. 암의 세부 종류에 따라 구분하여 치료하는 것이 필요함.
2) 생물지표(biomarker, e.g. HER-2)의 확인을 통한 특정 환자군을 선택하고 합당한 표적분자치료제를 적용하면 치료의 극대화를 꾀할 수 있다. 보다 나은 생물지표 검출기술의 개발 및 표준화가 필요함
3) 암의 heterogeneity로 인해 치료를 계속하다 보면 약제에 반응하지 않는 clone이 선택적으로 남게 되고 이로 인해 약제 내성이 발생할 수 있다. 다제병용요법(combination therapy) 도입으로 내성을 극복하고 임상성적을 보다 향상시킬 수 있다.
 8. Standard D2 lymph node dissection in minimally invasive surgery (Takahiro Kinoshita, National Cancer Center Hospital, East, Japan)
8. Standard D2 lymph node dissection in minimally invasive surgery (Takahiro Kinoshita, National Cancer Center Hospital, East, Japan)
국립암센터 동(東)병원에서는 위암수술의 75%가 복강경 수술입니다.
3 D CT simulation을 통하여 해부학적 구조를 다양한 각도에서 살펴볼 수 있어 큰 도움을 받고 있다고 합니다.
긴 동영상을 보았는데 피 한방울 나지 않았습니다. 강사는 "My goal is white surgery without any blood"라고 말하였습니다. White surgery. 멋진 말입니다.
Laparoscopic splenic hilar dissection without splenectomy 동영상은 상당히 인상적이었습니다.